Decreasing atomic radius
There will be an increase of ionization energy from left to right of a given period and a decrease from top to bottom. As one progresses down a group on the periodic table, the ionization energy will likely decrease, due to the greater number of shells, thereby positioning the valence electrons further from the protons, which attract them less, thereby requiring less energy to remove them. Trend-wise, the ionization potentials tend to increase while one progresses across a period because the greater number of protons (higher nuclear charge) attract the orbiting electrons more strongly, thereby increasing the energy required to remove one of the electrons. The first ionization energy is the energy required to remove one, the nth ionization energy is the energy required to remove the atom's nth electron, not including the n-1 electrons before it.

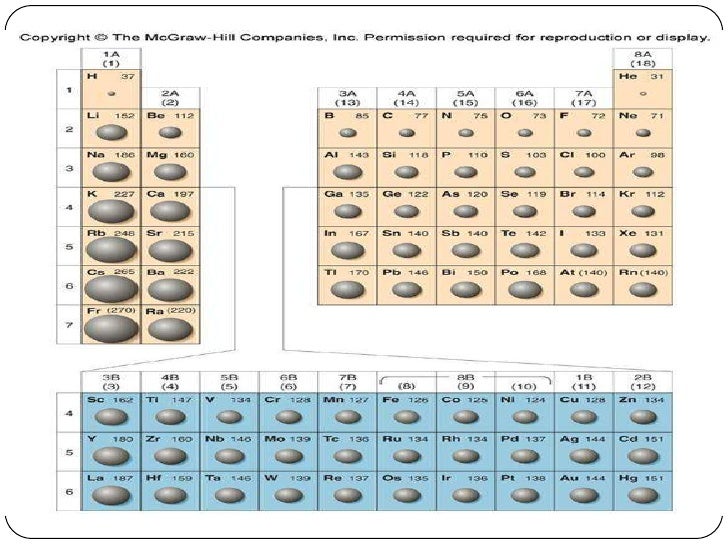
The ionization potential (or the ionization energy) is the miniumum energy required to remove one electron from each atom in a mole of atoms in the gaseous state. Atomic radii decrease left to right across a period. For example, lithium (145 pm) has a smaller atomic radius than magnesium (150 pm). However, diagonally, the number of protons has a larger effect than the sizeable radius. The atomic radius also will usually increase as one descends a group of the period table because the energy level (shell) increases down the group causing the outer shell electrons to be further away from the nucleus, thereby heavily increasing the atomic size. The atomic radius tends to decrease as one progresses across a period because the effective nuclear charge increases, thereby attracting the orbiting electrons and lessening the radius.

The atomic radius is the distance from the atomic nucleus to the outermost stable electron orbital in an atom that is at equilibrium. The Lewis acidity decreases going down however in a group as is evident to increasing metallic nature of the elements in group four. Elements to the right tend to have a greater way of attracting electrons then those on the left, making the left ones more basic whilst the right ones are acidic. This is due to the electronegativity of the elements. However, it decreases going down the table.
The Lewis acidity of each element tends to increase when moving from left to right in the periodic table.
